Quick positioning: what this glove is (and isn’t)
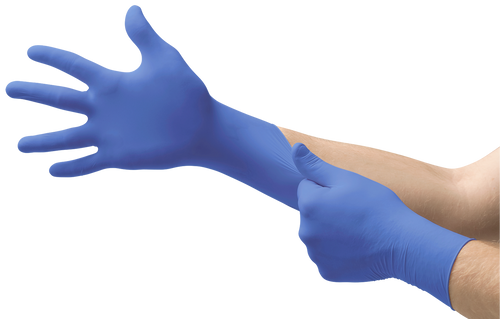
N19 is a non-sterile nitrile exam glove designed for broad, everyday use where texture-driven grip, consistent donning feel, and straightforward documentation support matter.
In controlled facilities, it most often lands in support zones, receiving/QC, maintenance, and general lab work — not in sterile core operations where sterile glove systems are required.
What customers use it for
- Sample taking and processing in lab support workflows.
- Equipment handling, maintenance, and repair work.
- Standard, moderate-risk examination procedures (site-defined).
- Food processing tasks with food-contact requirements (program-driven).
Why it stays on approved lists
- Texture that translates into control: fully textured surface supports more secure handling with tools and materials.
- Blue visibility: cobalt color is easy to spot in glove-change routines and compliance checks.
- Powder-free nitrile baseline: helps reduce powder-related contamination variables.
- Operational consistency: published thickness and AQL simplify comparisons across glove programs.
Build and composition notes
The manufacturer lists N19 as a nitrile exam glove with a fully textured external surface and antistatic property.
Typical thickness values are 0.10 mm (palm) and 0.11 mm (finger), with a typical overall length of 240 mm (9.5 in).
Specifications in context
| Attribute |
N19 (Ansell MICROFLEX® Cobalt®) |
| Material / color | Nitrile / cobalt blue |
| External surface | Fully textured |
| Powder content | Powder-free |
| Freedom from holes | 1.5 AQL (Inspection level I) |
| Length | 240 mm / 9.5 in (typical) |
| Thickness (typical) | Palm 0.10 mm / 3.9 mil; Finger 0.11 mm / 4.3 mil |
| Antistatic | Yes |
| Audit standard | EN ISO 13485:2012 |
| Country of origin | Malaysia |
| Packaging | 100/box; 10 boxes/case; 1,000/case |
| Chemo testing statement | Tested for use with chemotherapy drugs (ASTM D6978) and US FDA cleared |
Gowning education (ISO first): gloves are a contamination-control surface
- Donning sequence: gloves are typically donned late in the gowning sequence to keep the exterior surface cleaner.
- Touch discipline: once gloved, treat your hands as "product-side" surfaces; avoid face/hood adjustments and uncontrolled contacts.
- Change-out rules: define triggers (tear, wetting, task change, time) and train to them.
- Right glove for the zone: support-zone exam gloves are not a substitute for sterile cleanroom glove systems in critical areas.
EU GMP Annex 1 (after ISO): sterile-grade gowning expectations get stricter
EU GMP Annex 1 raises the bar for aseptic manufacture. For N19, the key point is placement: it can be an excellent support glove, while sterile/validated glove systems typically cover aseptic core work.
Practical handling habits that reduce glove-borne contamination
Small habits, big impact
- Handle by the cuff: minimize fingertip contact during donning.
- Avoid "glove drift": don't let gloved hands wander to phones, door handles, pens, or mask adjustments.
- Separate tasks: break "dirty handling" from "critical handling" with a glove change between them.
- Doff slowly: controlled removal reduces contamination spread.
Where programs commonly go wrong
- Wrong sizing: tears, fatigue, and loss of dexterity.
- Assuming "new" equals "clean": a glove can be contaminated immediately by poor donning/touch habits.
- Using non-sterile gloves in sterile zones: a qualification mismatch, not a training fix.
- Inconsistent glove-change triggers: different operators follow different rules without realizing it.
Why the Ansell ecosystem matters
Customers increasingly want PPE programs that are cohesive: gloves, garments, and eye/face protection that fit together operationally and administratively.
SOSCleanroom is building forward with this direction by expanding depth across Ansell-aligned lines so customers can standardize sourcing while keeping documentation and supply continuity tight.
Critical environment fit
N19 is typically best positioned in support functions and non-aseptic workflows where a robust nitrile exam glove is appropriate and sterility is not required.
SOSCleanroom note about SOP’s
The Technical Vault is written to improve product understanding and contamination-control technique. It is not your facility’s SOP, validation protocol, or regulatory interpretation.
Treat these notes as a starting point — then formalize what applies in your quality system.
Source basis
SOSCleanroom is the source for this Technical Vault entry.
Briefed and approved by the SOSCleanroom (SOS) staff.
If you have any questions please email us at Sales@SOSsupply.com
If you need additional information please try our SOSCleanroom specific AI ChatBot which draws from our extensive cleanroom specific libraries.
Last reviewed: May 1, 2026
© 2026 SOSCleanroom